Ghana leads in the approval of a novel malaria vaccine.
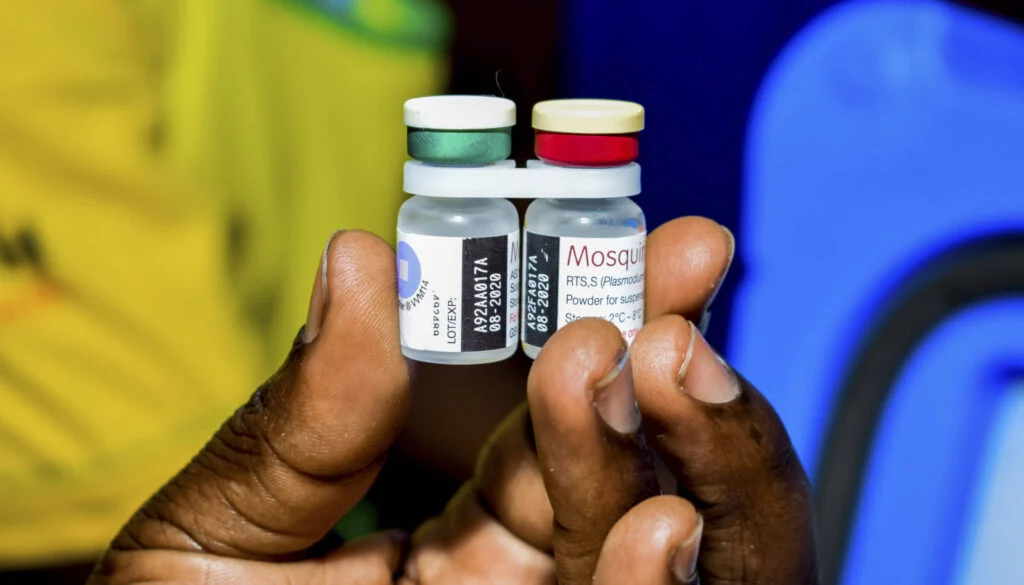

The final trial results for the novel malaria vaccine R21 have reportedly been evaluated by Ghana’s Food and Drugs Authority (FDA).
In comparison to earlier vaccines, the new one is reportedly much more effective.
James Gallagher, a health and science correspondent for the BBC, explains that despite the drug’s secrecy, the FDA plans to utilize it.
Children between the ages of five months and three will use it.

The trial’s approval by the World Health Organization (WHO) is also under consideration.
The director of the manufacturers Jenner Institute at the University of Oxford, Prof. Adrian Hill, is quoted as saying, “We expect R21 to make a major impact on malaria mortality in children in the coming years, and in the longer term [it] will contribute to overall final goal of malaria eradication and elimination.”
R21 is anticipated to cost a few bucks each dose.
The Serum Institute of India, which assisted AstraZeneca in developing the Covid-19 vaccinations, is getting ready to produce 100 to 200 million doses of the new malaria vaccine annually.
Additionally, the Institute plans to build a facility in Ghana to help with production.
According to CEO of the Serum Institute Adar Poonawalla, “developing a vaccine to greatly impact this huge disease burden has been extraordinarily difficult.”
The majority of the 620,000 people who die from malaria each year are very young toddlers.